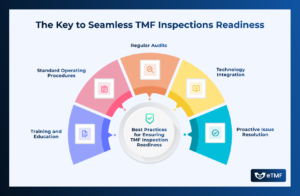
The Key to Seamless TMF Inspection Readiness
The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

When an inspection is declared for a study in your Trial Master File (TMF), thorough preparation is key to demonstrating compliance and operational excellence.

The Trial Master File (TMF) is a critical component in clinical trials, serving as the repository for all essential documents. TMF demonstrate the compliance
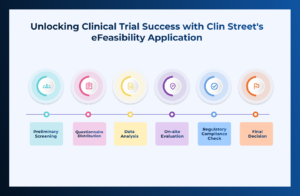
Ensuring that a site is capable of conducting a clinical trial is a pivotal first step in the landscape of Clinical Research! Clin Street’s

CLIN STREET LMS redefines training! It’s sleek, efficient, and packed with features to make administration a breeze. With a user-friendly interface and powerful tools,
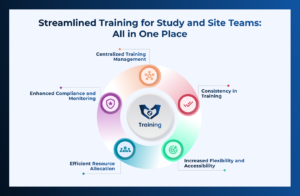
Compliance with regulatory requirements is paramount, in the world of Clinical Resarch. Organizations must adhere to guidelines set by bodies such as the US

The need for streamlined and efficient document management is more critical than ever. Enter CLIN STREET’s eTMF (electronic Trial Master File) application, a revolutionary

In the dynamic world of clinical research, where timelines are tight, regulations are stringent, and patient safety is paramount, effective milestone and event management

Events play a pivotal role in shaping trial outcomes, influencing patient safety, and determining the efficacy of investigational treatments. From adverse events to protocol
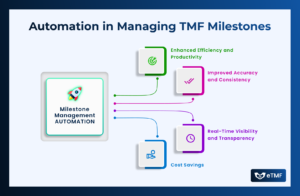
In the dynamic world of clinical research, Trial Master Files (TMFs) play a pivotal role in ensuring the integrity, completeness, and compliance of trial
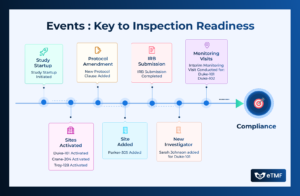
Clinical trials are dynamic endeavors characterized by a series of events, each playing a crucial role in the progression and success of the study.

Success of Clinical trials is critical to fulfill the hopes of real-life patients to receive better treatment options for their health condition. Every Clinical

Electronic Trial Master Files (eTMF) is gaining significant focus by regulatory agencies and has already become a central place to validate compliance of the

Adherence to regulatory standards is non-negotiable in Clinical Research. In the archival world, there is growing emphasis on achieving compliance. This blog serves as
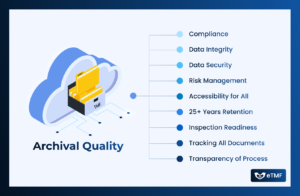
In clinical research, high-quality trial documentation drives not only fulfills the regulatory requirements but also ensures data integrity, patient safety, and research excellence. Archival

Maintaining compliance with regulatory standards is the most important piece in the conduct of clinical research to ensure the safety of participants and the
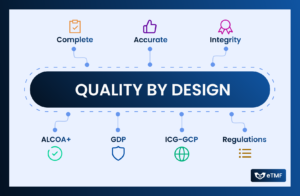
Clinical trials are complex in nature that demand meticulous attention to detail and adherence to stringent regulatory standards. At the heart of every successful
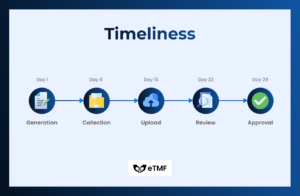
TMF timeliness refers to the promptness and efficiency with which Trial Master File (TMF) documents are collected, filed, updated, and maintained throughout the lifecycle

The integrity of the Trial Master File (TMF) is anchored in the ‘C’ of ‘ALCOA+’ principle known as “Contemporaneous“. In simple terms, Contemporaneous implies
As the world of clinical trials extends beyond borders, a globalized approach to Trial Master File (TMF) management becomes increasingly essential. CLIN STREET’s eTMF now

A Guide to Study TMF Archival In clinical trials, wrapping up a study is an important step that requires careful focus on details

In clinical trials, keeping information safe and controlling who can see it is really important. One aspect that adds complexity to this challenge is

At the core of every successful clinical trial is a meticulously organized and efficiently managed collection of critical documents, a responsibility entrusted to the
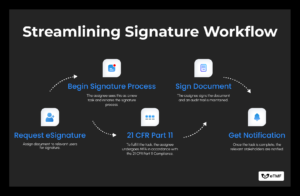
In clinical trials, the journey of a document involves scrutiny, collaboration, and, in many cases, obtaining signatures. CLIN STREET’s eTMF introduces an easy-to-use solution –

In clinical trials, the ability to maintain data integrity while streamlining processes is essential. CLIN STREET’s eTMF introduces the capability for site users to securely

In clinical trials, staying compliant and inspection-ready is critical. Failure to handle expiring documents efficiently can jeopardize inspection readiness and pose challenges for

In the fast-moving world of clinical trials, keeping your Trial Master File (TMF) in order is crucial. That’s where CLIN STREET’s eTMF comes in –

In the realm of clinical trials, an Electronic Trial Master File (eTMF) plays a pivotal role in ensuring the collection and management of clinical

If you’re an mNote user, you can take advantage of a convenient feature that allows you to create a website shortcut on your iPhone or

1. Visit app.clinstreet.com 2. Hit ‘Create an account’ 3. Input work email and details (No credit card required) 4. Log in to mNote

In the fast-paced world of clinical research, the reliance on Excel sheets for capturing visit notes may be holding you back. As the demands for

In the fast-paced world of clinical research, time is a valuable asset that can make or break success. For Clinical Research Associates (CRAs), efficient time

In the fast-paced world of clinical research, Clinical Research Associates (CRAs) are the vigilant guardians ensuring that every aspect of a clinical trial adheres to

In the dynamic field of clinical research, precision and accuracy are of utmost importance. Yet, some crucial aspects of the research process, such as visit

In the world of clinical research, keeping things organized and accessible is key. Every site and study contains crucial information, from contacts to associated people

In the field of clinical research, where accuracy holds utmost importance, the skill of revealing valuable insights and maneuvering through complex data intricacies stands as

In the dynamic world of clinical research, site visits stand as the cornerstone of a Clinical Research Associate’s (CRA) responsibilities. These visits hold a profound

In the fast-paced world of clinical research, precision and thoroughness are paramount. Clinical Research Associates (CRAs) play a pivotal role in ensuring the success of

In the fast-paced world of clinical research, staying organized and efficient during site visits is paramount. That’s where mNote steps in, revolutionizing the way Clinical