Managing Events : Ensuring Compliance and Success
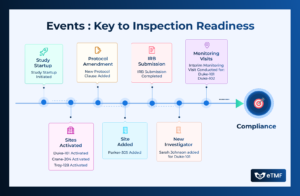
Clinical trials are dynamic endeavors characterized by a series of events, each playing a crucial role in the progression and success of the study.

Clinical trials are dynamic endeavors characterized by a series of events, each playing a crucial role in the progression and success of the study.

Success of Clinical trials is critical to fulfill the hopes of real-life patients to receive better treatment options for their health condition. Every Clinical

Electronic Trial Master Files (eTMF) is gaining significant focus by regulatory agencies and has already become a central place to validate compliance of the

Adherence to regulatory standards is non-negotiable in Clinical Research. In the archival world, there is growing emphasis on achieving compliance. This blog serves as
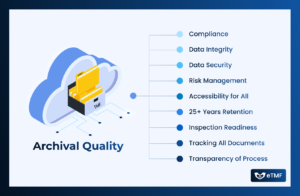
In clinical research, high-quality trial documentation drives not only fulfills the regulatory requirements but also ensures data integrity, patient safety, and research excellence. Archival
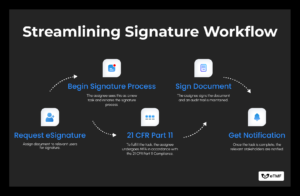
In clinical trials, the journey of a document involves scrutiny, collaboration, and, in many cases, obtaining signatures. CLIN STREET’s eTMF introduces an easy-to-use solution –