
Inspection Readiness: TMF Prep Essentials
When an inspection is declared for a study in your Trial Master File (TMF), thorough preparation is key to demonstrating compliance and operational excellence.

When an inspection is declared for a study in your Trial Master File (TMF), thorough preparation is key to demonstrating compliance and operational excellence.

The Trial Master File (TMF) is a critical component in clinical trials, serving as the repository for all essential documents. TMF demonstrate the compliance
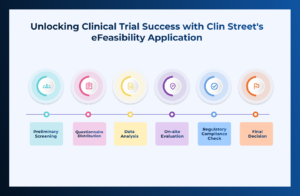
Ensuring that a site is capable of conducting a clinical trial is a pivotal first step in the landscape of Clinical Research! Clin Street’s

The need for streamlined and efficient document management is more critical than ever. Enter CLIN STREET’s eTMF (electronic Trial Master File) application, a revolutionary