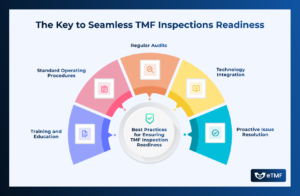
The Key to Seamless TMF Inspection Readiness
The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

When an inspection is declared for a study in your Trial Master File (TMF), thorough preparation is key to demonstrating compliance and operational excellence.
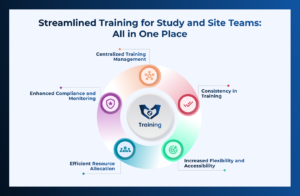
Compliance with regulatory requirements is paramount, in the world of Clinical Resarch. Organizations must adhere to guidelines set by bodies such as the US