The Key to Seamless TMF Inspection Readiness
The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

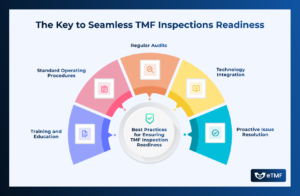
When an inspection is declared for a study in your Trial Master File (TMF), thorough preparation is key to demonstrating compliance and operational excellence.

The Trial Master File (TMF) is a critical component in clinical trials, serving as the repository for all essential documents. TMF demonstrate the compliance

In the dynamic world of clinical research, where timelines are tight, regulations are stringent, and patient safety is paramount, effective milestone and event management

Events play a pivotal role in shaping trial outcomes, influencing patient safety, and determining the efficacy of investigational treatments. From adverse events to protocol
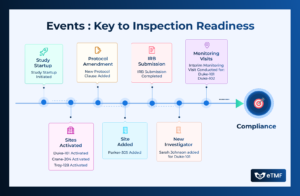
Clinical trials are dynamic endeavors characterized by a series of events, each playing a crucial role in the progression and success of the study.