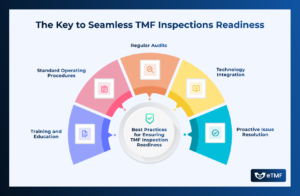
The Key to Seamless TMF Inspection Readiness
The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

The Trial Master File (TMF) serves as a comprehensive collection of essential documents that chronicle the planning, execution, and results of a study. Ensuring

When an inspection is declared for a study in your Trial Master File (TMF), thorough preparation is key to demonstrating compliance and operational excellence.

The Trial Master File (TMF) is a critical component in clinical trials, serving as the repository for all essential documents. TMF demonstrate the compliance
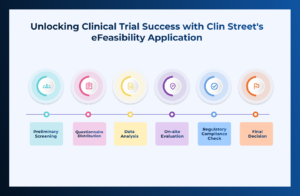
Ensuring that a site is capable of conducting a clinical trial is a pivotal first step in the landscape of Clinical Research! Clin Street’s

CLIN STREET LMS redefines training! It’s sleek, efficient, and packed with features to make administration a breeze. With a user-friendly interface and powerful tools,
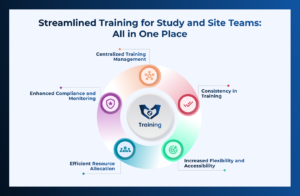
Compliance with regulatory requirements is paramount, in the world of Clinical Resarch. Organizations must adhere to guidelines set by bodies such as the US

The need for streamlined and efficient document management is more critical than ever. Enter CLIN STREET’s eTMF (electronic Trial Master File) application, a revolutionary

In the dynamic world of clinical research, where timelines are tight, regulations are stringent, and patient safety is paramount, effective milestone and event management

Events play a pivotal role in shaping trial outcomes, influencing patient safety, and determining the efficacy of investigational treatments. From adverse events to protocol
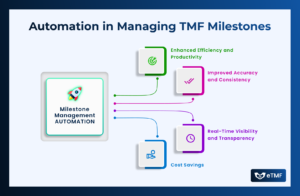
In the dynamic world of clinical research, Trial Master Files (TMFs) play a pivotal role in ensuring the integrity, completeness, and compliance of trial